




Abstract
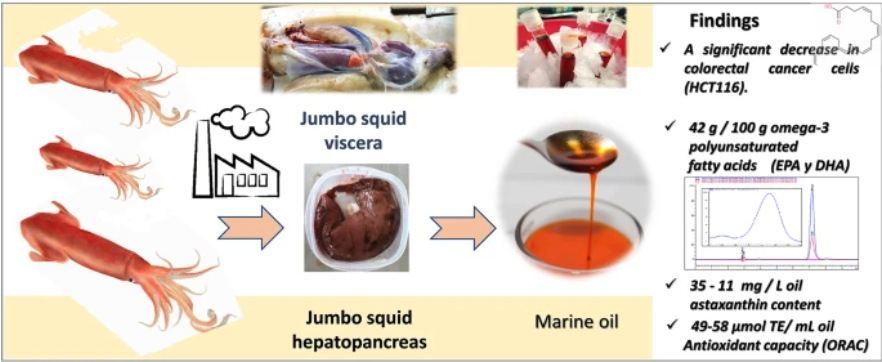
The global impact of residues resulting from fishing processing has generated the need to develop sustainable processes, find additional potential of raw materials, and provide alternatives for their recovery of potentially valuable by-products. On the other hand, colorectal cancer (CRC) is the second leading cause of cancer death worldwide and needs novel and more effective treatments. In this study, we extracted and characterized oil from Jumbo squid hepatopancreas and evaluated its antioxidant potential and antiproliferative activity. The oil recovered from the Jumbo squid production process has characteristics that meet international standards. PUFA-rich oil from Jumbo squid hepatopancreas showed high concentrations of EPA/DHA (about 42% of the total fatty acids) and antioxidant potential. The antiproliferative activity evaluated on colorectal cancer cells, HCT116, showed activity against the tumor cells, representing the first evidence for a potential anticancer application. This research proposes a sustainable strategy for achieving a circular bioeconomy by recovering oil from jumbo squid viscera, offering alternatives for valorizing this bioproduct.

Abstract
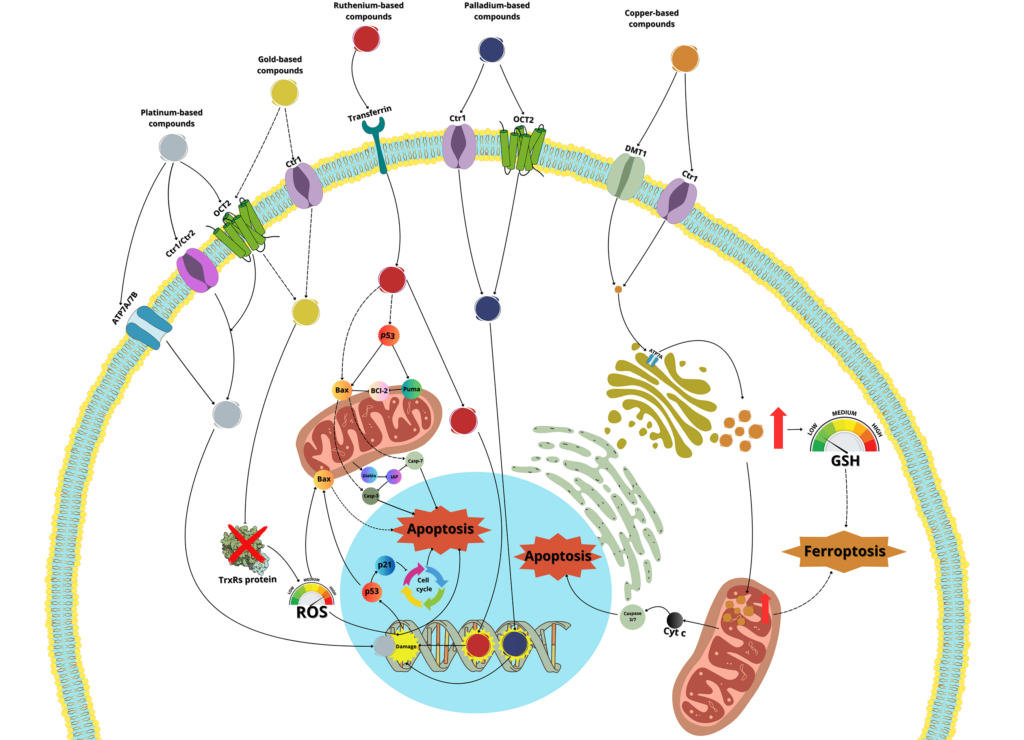
Cancer remains one of the leading causes of death in the world, with more than 9 million deaths in 2022, a number that continues to rise. This highlights the urgent need for the development of new drugs, with enhanced antitumor capabilities and fewer side effects. Metal-based drugs have been used in clinical practice since the late 1970s, beginning with the introduction of cisplatin. Later, two additional platinum-based molecules, carboplatin, and oxaliplatin, were introduced, and all three continue to be widely used in the treatment of various cancers. However, despite their significant anticancer activity, the undesirable side effects of these drugs have motivated the scientific community to explore other metal-based complexes with greater anticancer potential and fewer adverse effects. In this context, metals such as ruthenium, copper, gold, zinc, palladium, or iridium, present promising alternatives for the development of new anticancer agents. Unfortunately, although thousands of metal-based drugs have been synthesized and tested both in vitro and in animal models, only a few ruthenium-based drugs have entered clinical trials in recent years. Meanwhile, many other molecules with comparable or even greater anticancer potential have not advanced beyond the laboratory stage. In this review, we will revisit the mechanisms of action and anticancer activities of established platinum-based drugs and explore their use in recent clinical trials. Additionally, we will examine the development of potential new metal-based drugs that could one day contribute to cancer treatment worldwide.
Key words: Metal-based drugs, Cancer treatment, Chemotherapy

Abstract
Background: Tobacco use is one of the main risk factors for Lung Cancer (LC) development. However, about 10-20% of those diagnosed with the disease are never-smokers. For Non-Small Cell Lung Cancer (NSCLC) there are clear differences in both the clinical presentation and the tumor genomic profiles between smokers and never-smokers. For example, the Lung Adenocarcinoma (LUAD) histological subtype in never-smokers is predominately found in young women of European, North American, and Asian descent. While the clinical presentation and tumor genomic profiles of smokers have been widely examined, never-smokers are usually underrepresented, especially those of a Latin American (LA) background. In this work, we characterize, for the first time, the difference in the genomic profiles between smokers and never-smokers LC patients from Chile.
Methods: We conduct a comparison by smoking status in the frequencies of genomic alterations (GAs) including somatic mutations and structural variants (fusions) in a total of 10 clinically relevant genes, including the eight most common actionable genes for LC (EGFR, KRAS, ALK, MET, BRAF, RET, ERBB2, and ROS1) and two established driver genes for malignancies other than LC (PIK3CA and MAP2K1). Study participants were grouped as either smokers (current and former, n = 473) or never-smokers (n = 200) according to self-report tobacco use at enrollment.
Results: Our findings indicate a higher overall GA frequency for never-smokers compared to smokers (58 vs. 45.7, p-value < 0.01) with the genes EGFR, KRAS, and PIK3CA displaying the highest prevalence while ERBB2, RET, and ROS1 the lowest. Never-smokers present higher frequencies in seven out of the 10 genes; however, smokers harbor a more complex genomic profile. The clearest differences between groups are seen for EGFR (15.6 vs. 21.5, p-value: < 0.01), PIK3CA (6.8 vs 9.5) and ALK (3.2 vs 7.5) in favor of never-smokers, and KRAS (16.3 vs. 11.5) and MAP2K1 (6.6 vs. 3.5) in favor of smokers. Alterations in these genes are comprised almost exclusively by somatic mutations in EGFR and mainly by fusions in ALK, and only by mutations in PIK3CA, KRAS and MAP2K1.
Conclusions: We found clear differences in the genomic landscape by smoking status in LUAD patients from Chile, with potential implications for clinical management in these limited-resource settings.
Keywords: Cancer disparities; Chilean; EGFR mutation; KRAS; Latin American populations; Lung Adenocarcinoma; MAP2K1; Tobacco consumption.
